Role of Hepcidin in Anemia of Chronic Disease
Authors
Files
Abstract
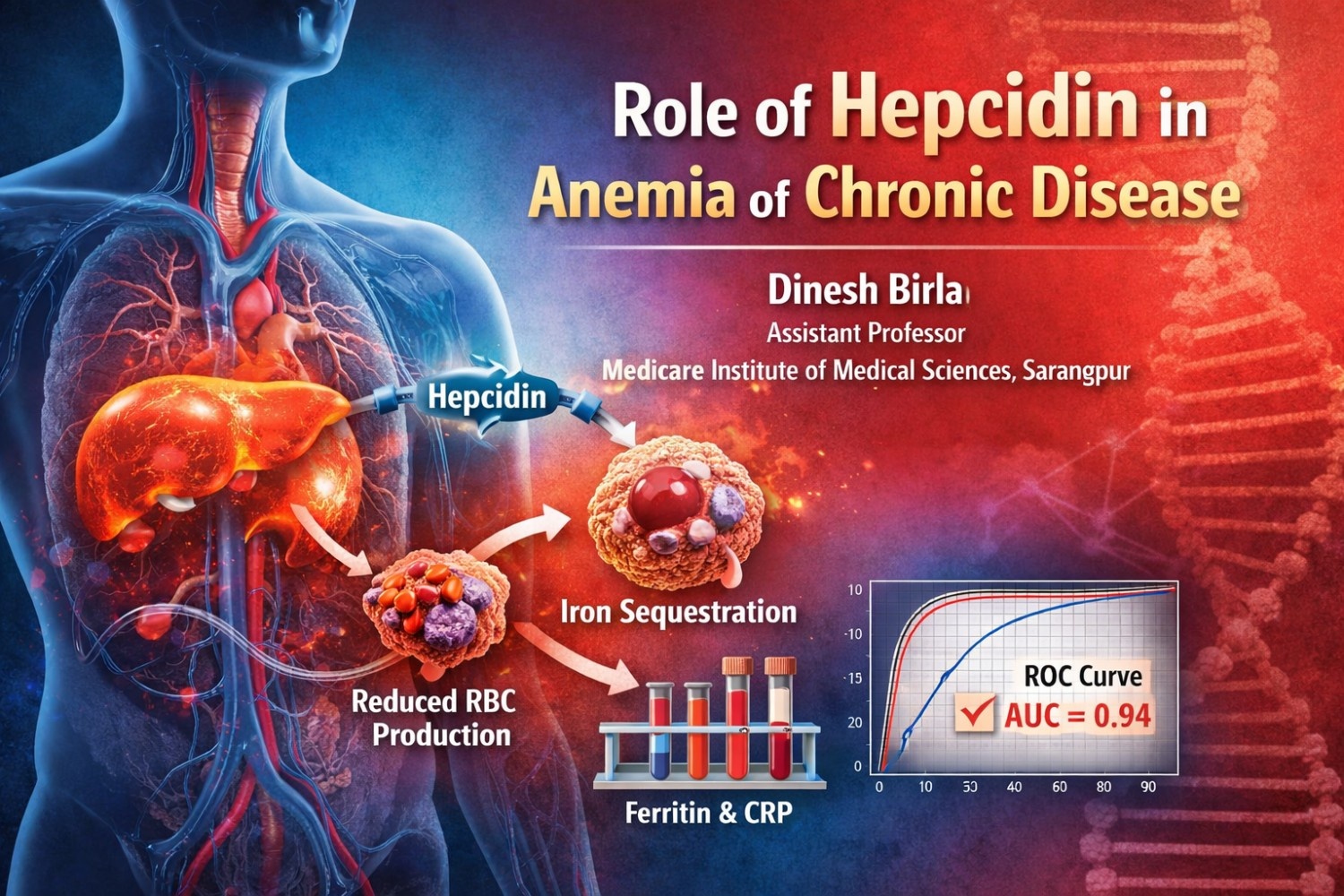
Anemia of Chronic Disease (ACD), also known as anemia of inflammation, is the second most common cause of anemia worldwide and is commonly observed in patients with chronic infections, autoimmune disorders, malignancies, and chronic kidney disease. Unlike Iron Deficiency Anemia (IDA), ACD is characterized by adequate iron stores but impaired iron utilization. Hepcidin, a hepatic peptide hormone, plays a central role in systemic iron regulation and is considered a key factor in the pathogenesis of ACD. The present hospital-based analytical case control study included 150 participants divided into three groups: 50 patients with ACD, 50 with IDA, and 50 healthy controls. All participants underwent complete blood count, serum iron profile, serum ferritin, C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and serum hepcidin estimation using the sandwich nitrocellulose membrane technique. Statistical analysis was performed using SPSS version 25.0. Group comparisons were carried out using ANOVA and independent t-tests, correlations were assessed using Pearson’s correlation coefficient, and diagnostic accuracy was evaluated through Receiver Operating Characteristic (ROC) curve analysis. The results showed that mean serum hepcidin levels were significantly higher in the ACD group (62.4 ± 15.8 ng/mL) compared to the IDA group (8.9 ± 4.2 ng/mL) and healthy controls (21.6 ± 7.4 ng/mL), with p < 0.001. A strong positive correlation was observed between serum hepcidin and ferritin (r = 0.72, p < 0.001) as well as CRP (r = 0.69, p < 0.001), while a significant negative correlation was found between serum hepcidin and hemoglobin levels (r = −0.58, p < 0.001). ROC curve analysis demonstrated excellent diagnostic performance of serum hepcidin in differentiating ACD from IDA, with an area under the curve (AUC) of 0.94, sensitivity of 92%, and specificity of 88% at a cut-off value of 32 ng/mL. The findings confirm that elevated serum hepcidin plays a crucial role in the development of ACD through inflammation-driven iron sequestration and impaired erythropoiesis. Serum hepcidin shows high diagnostic accuracy in distinguishing ACD from IDA and may serve as a valuable clinical biomarker, although larger studies are needed to establish standardized reference ranges and explore therapeutic strategies targeting hepcidin modulation.
References
1. Guralnik, J. M., Eisenstaedt, R. S., Ferrucci, L., Klein, H. G., & Woodman, R. C. (2004). Prevalence of anemia in persons 65 years and older in the United States: evidence for a high rate of unexplained anemia. Blood, 104(8), 2263–2268. https://doi.org/10.1182/blood-2004-05-1812
2. Alemayehu, M. (2017). Factors Associated with Anemia among Lactating Mothers in Subsistence Farming Households from Selected Districts of Jimma Zone, South Western Ethiopia: A Community Based Cross-Sectional Study. Journal of Nutrition & Food Sciences, 07(03). https://doi.org/10.4172/2155-9600.1000595
3. Wiciński, M., Liczner, G., Cadelski, K., Kołnierzak, T., Nowaczewska, M., & Malinowski, B. (2020). Anemia of Chronic Diseases: Wider Diagnostics—Better Treatment? Nutrients, 12(6), 1784. https://doi.org/10.3390/nu12061784
4. Schubert, T., Echtenacher, B., Hofstädter, F., & Männel, D. N. (2003). TNF-Independent development of transient anemia of chronic disease in a mouse model of protracted septic peritonitis. Laboratory Investigation, 83(12), 1743–1750. https://doi.org/10.1097/01.lab.0000101693.12149.2c
5. Raj, R., Menon, N., & Srivaths, L. (2025). Iron deficiency and anemia in menstruating adolescents. Pediatrics in Review, 46(11), 606–616. https://doi.org/10.1542/pir.2025-006756
6. Boutou, A. K., Stanopoulos, I., Pitsiou, G. G., Kontakiotis, T., Kyriazis, G., Sichletidis, L., & Argyropoulou, P. (2011). Anemia of Chronic Disease in Chronic Obstructive Pulmonary Disease: A Case-Control Study of Cardiopulmonary Exercise Responses. Respiration, 82(3), 237–245. https://doi.org/10.1159/000326899
7. Brown, K., Glen, S., & White, T. (1987). LOW SERUM IRON STATUS AND AKATHISIA. The Lancet, 329(8544), 1234–1236. https://doi.org/10.1016/s0140-6736(87)92687-0
8. Döring, G., & Hoiby, N. (2004). Early intervention and prevention of lung disease in cystic fibrosis: a European consensus. Journal of Cystic Fibrosis, 3(2), 67–91. https://doi.org/10.1016/j.jcf.2004.03.008
9. Schwartz, S. I., & Brunicardi, F. C. (2010). Schwartz’s Principles of Surgery, Ninth Edition. McGraw Hill Professional.
10. Aapro, M., Beguin, Y., Bokemeyer, C., Dicato, M., Gascón, P., Glaspy, J., Hofmann, A., Link, H., Littlewood, T., Ludwig, H., Österborg, A., Pronzato, P., Santini, V., Schrijvers, D., Stauder, R., Jordan, K., & Herrstedt, J. (2017). Management of anaemia and iron deficiency in patients with cancer: ESMO Clinical Practice Guidelines. Annals of Oncology, 29(Suppl 4), iv96–iv110. https://doi.org/10.1093/annonc/mdx758
11. Macciò, A., & Madeddu, C. (2012). Management of anemia of inflammation in the elderly. Anemia, 2012, 1–20. https://doi.org/10.1155/2012/563251
12. Aschemeyer, S. (2017). The mechanism of action and regulation of Hepcidin.
13. Fosburg, M., & Nathan, D. (1990). Treatment of Cooley’s anemia [see comments]. Blood, 76(3), 435–444. https://doi.org/10.1182/blood.v76.3.435.435
14. Wick, M., Pinggera, W., & Lehmann, P. (2000). Iron Metabolism, Anemias, diagnosis, and therapy: Novel Concepts in the Anemias of Renal and Rheumatoid Disease. Springer Science & Business Media.
15. Iron-Deficiency Anemia: New Insights for the Healthcare Professional: 2011 edition: ScholarlyPaper. (2012). ScholarlyEditions.
16. Jowett, S. L., Seal, C. J., Pearce, M. S., Phillips, E., Gregory, W., Barton, J. R., & Welfare, M. R. (2004). Influence of dietary factors on the clinical course of ulcerative colitis: a prospective cohort study. Gut, 53(10), 1479–1484. https://doi.org/10.1136/gut.2003.024828
17. Salie, R. (2005). Mouse RGMs : a three protein family with diverse function and localization. In edoc (University of Basel). https://doi.org/10.5451/unibas-004021941
18. Nicolas, G., Chauvet, C., Viatte, L., Danan, J. L., Bigard, X., Devaux, I., Beaumont, C., Kahn, A., & Vaulont, S. (2002). The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. Journal of Clinical Investigation, 110(7), 1037–1044. https://doi.org/10.1172/jci0215686
19. Arora, S. (2012). Iron metabolism. BoD – Books on Demand.
20. Joshi, M., Adhikari, B., Aldred, P., Panozzo, J., Kasapis, S., & Barrow, C. (2012). Interfacial and emulsifying properties of lentil protein isolate. Food Chemistry, 134(3), 1343–1353. https://doi.org/10.1016/j.foodchem.2012.03.029
21. Reichert, C. O. (2018). Influence of hepcidin in the development of anemia.
22. Kimball, E. S. (2024). Cytokines and inflammation. CRC Press.
23. Eddowes, L. A. (2010). Interactions of HIV-1 and Hepatitis C Virus with Iron Metabolism.
24. Gkouvatsos, K. (2013). Insights on the role of the protein hemojuvelin in the maintenance of systemic iron homeostasis.




